
Concept
The foundation of living systems is based on chemical reactions. They are spatiotemporally regulated in response to extra- or intracellular stimuli. Of importance to understand the mechanism of life is the ability to visualize and to perturb these reactions. Chemical tools provide unique means to emulate or modulate biological systems to either investigate the underlying biology or create new function.
In my laboratory, we run both biology-driven and chemistry-driven projects. In the former, we have defined biological questions, which are not readily solved by traditional biological methods. We tackle these problems using novel chemical approaches in combination with biochemical and cell biological methods. In the later, we develop novel chemical tools that can be widely used to investigate biology. These approaches make it possible to reveal new mechanism of biology.
Autophagy
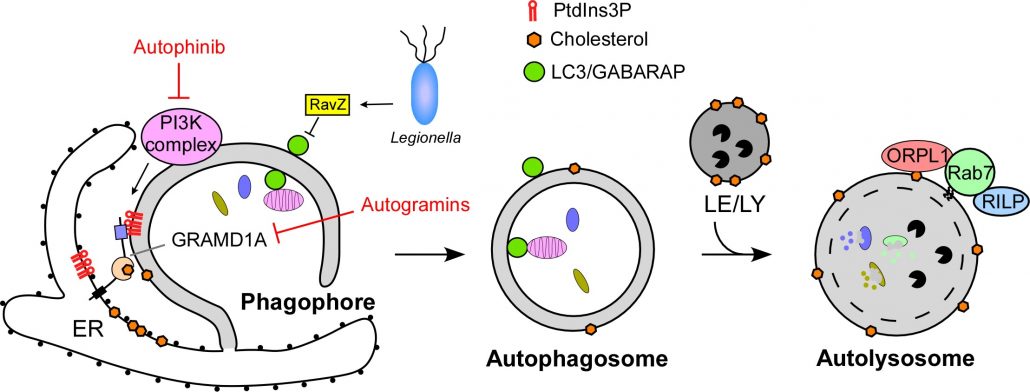
Autophagy is an evolutionarily conserved self-eating process in eukaryotes, mainly involved in elimination of organelles and aggregated proteins. Malfunction of autophagy has been associated with diverse human diseases, including cancer, neurodegeneration, cardiac hypertrophy, and pathogen infection. Formation of double-membrane autophagosome is the key process in autophagy. The regulatory pathways of autophagy and the formation of autophagosome remain the most important questions for understanding autophagy.
Using a combination of cell biology, structural biology and chemical genetic approaches, we have elucidated fundamental mechanisms underlying autophagy. Our laboratory has elaborated a novel mode of action for virulent bacteria (Legionella) against host autophagy (eLife 2017). We have identified novel chemotypes for autophagy modulation and new cellular targets involved in new mechanism of autophagy regulation (Angew Chem 2017, Nat Chem Biol 2019).
Membrane trafficking and small GPTases (G-proteins)
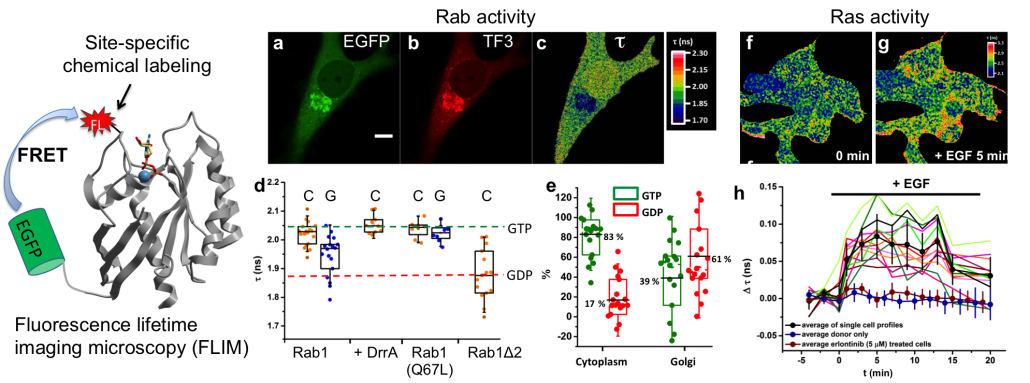
Due to the spatial segregation of intracellular organelles of eukaryotic cells a sophisticated system of vesicular transport is required to ensure the communication between different compartments. Rab GTPases function as key regulators of intracellular vesicular transport. With the help a complex network of Rab regulators and effectors, Rab GTPases regulate these processes through tightly controlled enzymatic GTPase cycle and spatial distribution in cells. We are interested in answering the question of how these complex biological reactions are coordinated and how the malfunction of these processes link to diseases. These questions have been assessed at both the molecular and the cellular level in a quantitative manner (Nat Chem Biol 2010, JACS 2012, PNAS 2014, PNAS 2016, Biochem 2019).
Protein chemical modification
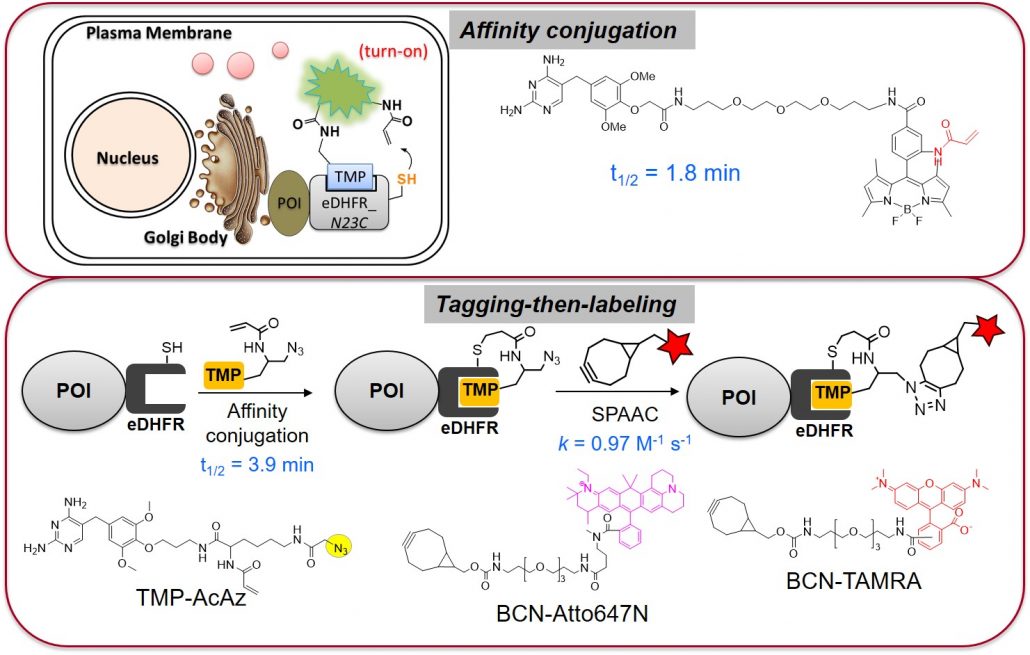
Labeling proteins with synthetic probes, such as fluorophores, affinity tags, and other functional labels is enormously useful for characterizing protein function in vitro, in live cells, or in whole organisms. Chemical methods have substantially expanded the tools that are can be used to modify proteins. Our work has been focused on preparation of post-translationally modified proteins (ChemBioChem 2011, 2013, Bioorg Med Chem 2017). These semi-synthetic protein probes have been very useful in the investigation of protein functions in membrane trafficking and autophagy. We are interested in developing new methods for protein modification/labeling (Angew Chem 2011, JACS 2014, Chem Comm 2015, Angew Chem 2016). These methodologies have been valuable for visualizing biochemical reactions in living systems and proteomic studies. Such developments not only facilitate the study of the system we are interested but contribute new tools to the larger biological field.
Chemical and chemo-optogenetics
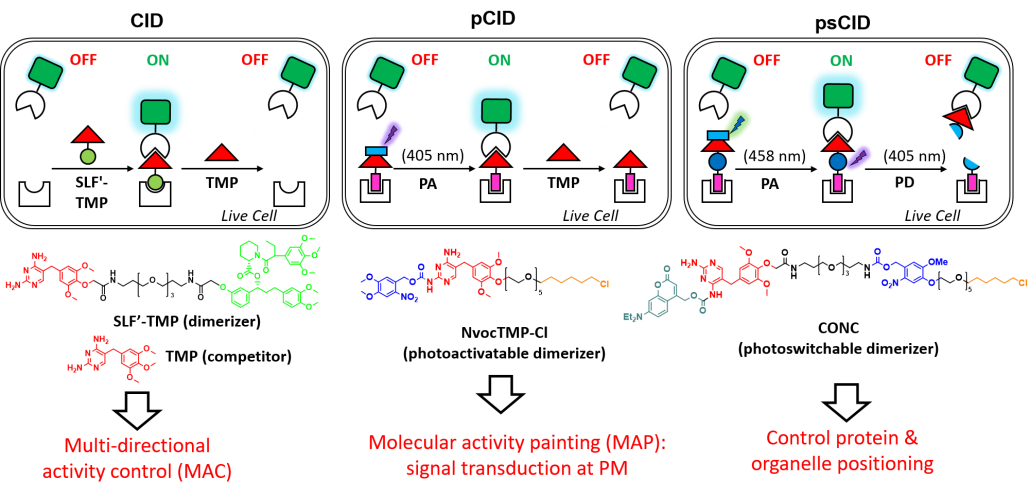
Genetic perturbations by gain or loss of gene function through overexpression, knock-out or knock-down approaches are powerful for biological studies. However, traditional genetic approaches are chronic (hours to days). Consequently, the phenotype may not be detected due to adaptation and the dynamics of phenotypic change cannot be followed. Chemical genetic approaches using small molecules are acute, reversible, conditional and tunable and have been very useful to dissect the complexity of biological regulatory networks. However, many of these compounds have additional off-target effects that may confound elucidation of biological systems in certain contexts. Our laboratory has developed the first bioorthogonal and reversible chemically induced dimerization (CID) system (SLF’-TMP system) for controlling protein function by small molecules in live cells (Angew Chem 2014). A photoactivatable CID (pCID) and photo-switchable CID system (psCID) using photo-caged dimerizer (NovcTMP-Cl) (Angew Chem 2017) and photo-switchable dimerizer (CONC) allow superior spatiotemporal control of protein function in live cells (Angew Chem 2018). We have developed novel “Molecular Activity Painting (MAP)” strategy by combining pCID with immobilized artificial receptors to enable “painting” of signaling molecules and their activity at micrometer-scale at the plasma membrane. Using photoactivatable dual-chemically induced dimerization (pdCID) system, we developed a Multi-directional Activity Control (MAC) approach to spatiotemporally control cellular signaling and intracellular cargo transport (Angew Chem 2018).
Selected recent publications:
1. Laraia L, Friese A, Corkery DP, Konstantinidis G, Erwin N, Hofer W, Karatas H, Klewer L, Brockmeyer A, Metz M, Schölermann B, Dwivedi M, Li L, Rios-Munoz P, Köhn M, Winter R, Vetter IR, Ziegler S, Janning P, Wu YW*, Waldmann H*. (2019) The cholesterol transfer protein GRAMD1A regulates autophagosome biogenesis. Nat. Chem. Biol. 15(7):710-720.
2. Chen X, Venkatachalapathy M, Dehmelt L, Wu YW*. (2018) Multidirectional Activity Control of Cellular Processes by a Versatile Chemo-optogenetic Approach. Angew. Chem. Int. Ed. 57(37):11993-11997.
3. Yang A, Pantoom S, Wu YW*. (2017) Elucidation of anti-autophagy mechanism of the Legionella effector RavZ using semisynthetic LC3 proteins. eLife pii: e23905.
4. Voss S, Krüger DM, Koch O, Wu YW*. (2016) Spatiotemporal imaging of small GTPases activity in live cells. Proc. Natl. Acad. Sci. U. S. A. 113(50):14348-14353.
5. Liu W, Li F, Chen X, Yi L, Wu YW*. (2014) A rapid and fluorogenic TMP-AcBOPDIPY probe for covalent labeling of proteins in live cells. J. Am. Chem. Soc. 136 (12): 4468-71.

