Mechanism of enzyme activation discovered
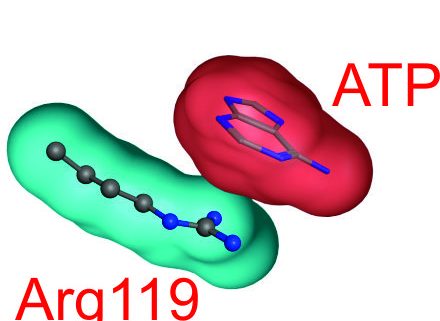
In an ISB effort the research groups lead by Anna Linusson, Elisabeth Sauer-Eriksson and Magnus Wolf-Watz has discovered a key event in activation of the essential enzyme adenylate kinase. It was discovered that the large-scale and activating conformational change triggered by ATP binding is nucleated by a strong cation-PI interaction formed between the cationic sidechain of an arginine with the aromatic adenosine base of ATP. The discovery may pave way for future enzyme design efforts where recognition of aromatic systems is required. The finding was made possible through an integrative effort using DFT calculations, NMR spectroscopy and x-ray crystallography. The team consisted of Per Rogne, David Andersson, Christin Grundström, Elisabeth Sauer-Eriksson, Anna Linusson and Magnus Wolf-Watz. The finding is published in Biochemistry https://pubs.acs.org/doi/10.1021/acs.biochem.9b00538.